Protein Motional Details Revealed by Complementary Structural Biology Techniques
2020-06-23
Kristof Grohe, Snehal Patel, Cornelia Hebrank, Sara Medina, Alexander Klein, Petra Rovó, Suresh K. Vasa, Himanshu Singh, Beat Vögeli, Lars V. Schäfer, and Rasmus Linser
Structure, 2020, 28, p 1024–2034
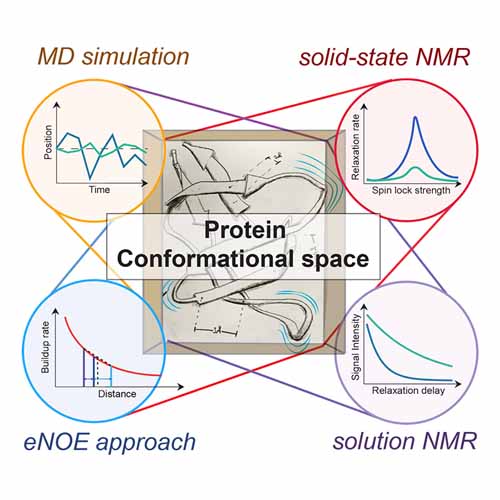
Proteins depend on defined molecular plasticity for their functionality. How to comprehensively capture dynamics correctly is of ubiquitous biological importance. Approaches commonly used to probe protein dynamics include model-free elucidation of site-specific motion by NMR relaxation, molecular dynamics (MD)-based approaches, and capturing the substates within a dynamic ensemble by recent eNOE-based multiple- structure approaches. Even thoughMDis sometimes combinedwith ensemble-averagedNMR restraints, these approaches have largely been developed and used individually. Owing to the different underlying concepts and practical requirements, it has remained unclear how they compare, and how they cross-validate and complement each other. Here, we extract and compare the differential information contents of MD simulations, NMR relaxationmeasurements, and eNOE-based multi-state structures for the SH3 domain of chicken a-spectrin. The data show that a validated, consistent, and detailed picture is feasible both for timescales and actual conformational states sampled in the dynamic ensemble. This includes the biologically important sidechain plasticity, for which experimentally cross-validated assessment is a significant challenge.