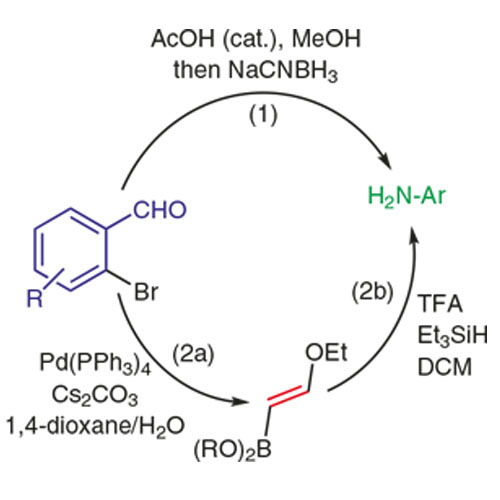
A Short Approach to N-Aryl-1,2,3,4-tetrahydroisoquinolines from N-(2-Bromobenzyl)anilines via a Reductive Amination/Palladium- Catalyzed Ethoxyvinylation/Reductive N-Alkylation Sequence
2021-01-07
Carina Glas, Ricky Wirawan, Franz Bracher
N-Aryl-1,2,3,4-tetrahydroisoquinolines are obtained via a convenient and short protocol with a broad range of substituents on both aromatic rings and high functional group tolerance. Starting from readily available ortho-brominated aromatic aldehydes and primary aromatic amines, condensation of these building blocks under reductive conditions gives N-aryl 2-bromobenzylamines. The C-3/C-4-unit of the tetrahydroisoquinoline is introduced using commercially available 2-ethoxyvinyl pinacolboronate under Suzuki conditions. Finally, the obtained crude ortho-ethoxyvinyl benzylamines are cyclized via an intramolecular reductive amination using the combination of triethylsilane/TFA to give the desired N-aryl-1,2,3,4-tetrahydroisoquinolines.