RNA-Templated Peptide Bond Formation Promotes L-Homochiralty
2024-02-26
Ewa Węgrzyn, Dr. Ivana Mejdrová, Dr. Felix M. Müller, Dr. Milda Nainytė, Dr. Luis Escobar, Prof. Dr. Thomas Carell
Angewandte Chemie, Volume 63, Issue 19, e202319235, 2024
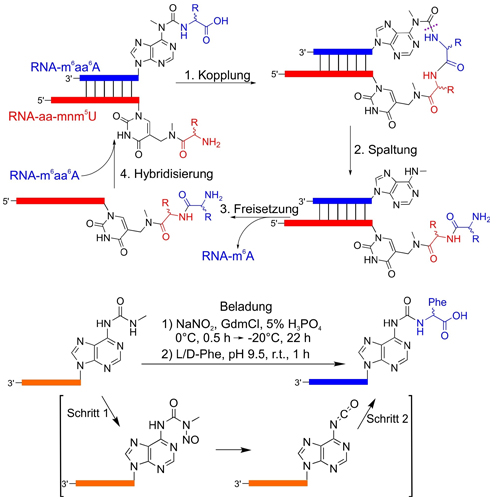
The world in which we live is homochiral. The ribose units that form the backbone of DNA and RNA are all D-configured and the encoded amino acids that comprise the proteins of all living species feature an all-L-configuration at the α-carbon atoms. The homochirality of α-amino acids is essential for folding of the peptides into well-defined and functional 3D structures and the homochirality of D-ribose is crucial for helix formation and base-pairing. The question of why nature uses only encoded L-α-amino acids is not understood. Herein, we show that an RNA-peptide world, in which peptides grow on RNAs constructed from D-ribose, leads to the self-selection of homo-L-peptides, which provides a possible explanation for the homo-D-ribose and homo-L-amino acid combination seen in nature.