5-Methylcytosine is Oxidized to the Natural Metabolites of TET Enzymes by a Biomimetic Iron(IV)-Oxo Complex
2019-06-18
Niko S. W. Jonasson and Lena J. Daumann
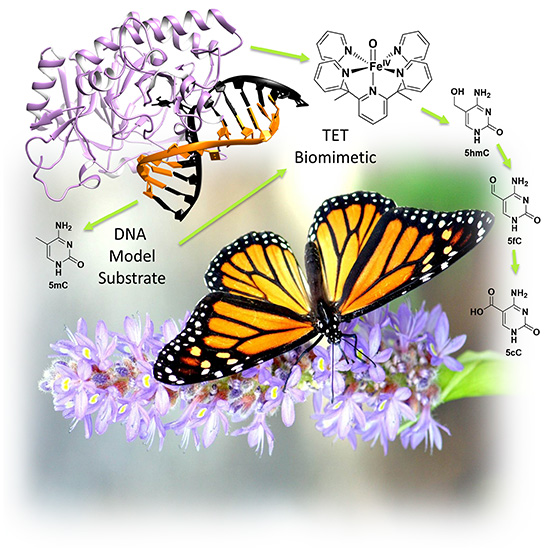
Ten‐eleven‐translocation (TET) methyl cytosine dioxygenases play a key role in epigenetics by oxidizing the epigenetic marker 5‐methyl cytosine (5mC) to 5‐hydroxymethyl cytosine (5hmC), 5‐formyl cytosine (5fC), and 5‐carboxy cytosine (5cC). Although much of the metabolism of 5mC has been studied closely, certain aspects—such as discrepancies among the observed catalytic activity of TET enzymes and calculated bond dissociation energies of the different cytosine substrates—remain elusive. Here, it is reported that the DNA base 5mC is oxidized to 5hmC, 5fC, and 5cC by a biomimetic iron(IV)‐oxo complex, reminiscent of the activity of TET enzymes. Studies show that 5hmC is preferentially turned over compared with 5mC and 5fC and that this is in line with the calculated bond dissociation energies. The optimized syntheses of d3‐5mC and d2‐5hmC are also reported and in the reaction with the biomimetic iron(IV)‐oxo complex these deuterated substrates showed large kinetic isotope effects, confirming the hydrogen abstraction as the rate‐limiting step. Taken together, these results shed light on the intrinsic reactivity of the C−H bonds of epigenetic markers and the contribution of the second coordination sphere in TET enzymes.